Introduction: When a Vascular Drug Learns Immunology
Medicine has a long tradition of underestimating its own discoveries. A compound is developed with one purpose in mind, proves effective, and is promptly placed into a neat clinical box. Then, years later, someone asks an inconvenient question: “What else does it do?” This is precisely the story of phosphodiesterase-5 (PDE-5) inhibitors.
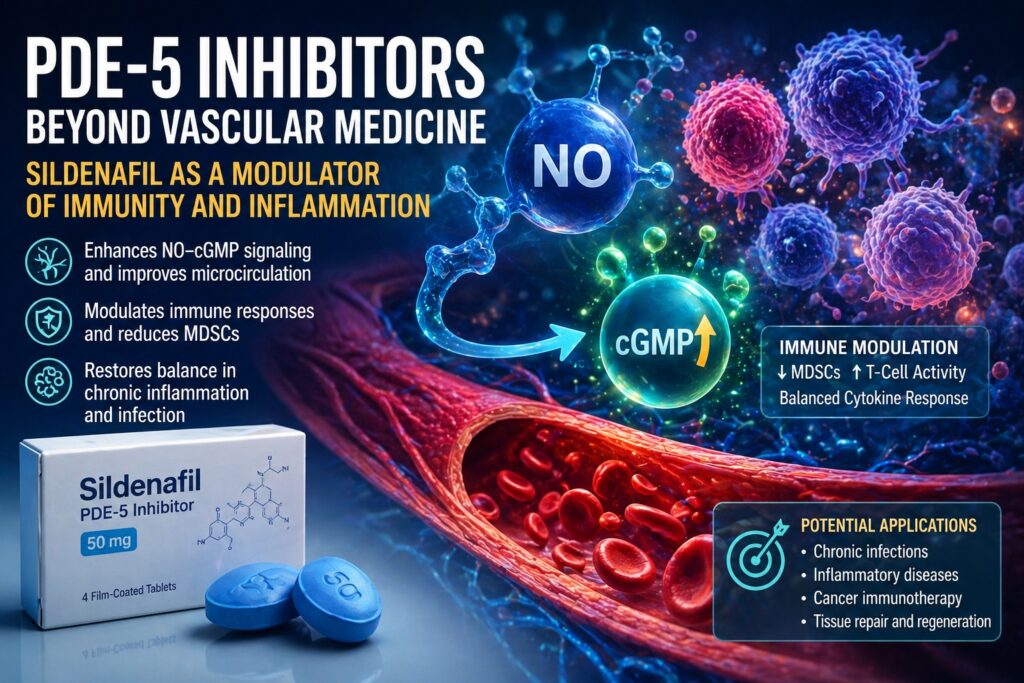
Initially introduced as targeted vasodilators, PDE-5 inhibitors—most notably sildenafil—quickly gained global recognition for their role in treating erectile dysfunction. Yet their pharmacological reach extends far beyond the vascular beds of the corpus cavernosum. These agents interact with one of the most fundamental signaling pathways in human physiology: the nitric oxide (NO)–cyclic guanosine monophosphate (cGMP) axis.
Recent immunological research reveals that PDE-5 inhibitors influence not only vascular tone but also immune cell behavior, inflammatory responses, and tissue homeostasis. The implications are profound. What was once a niche therapy is now emerging as a systemic modulator of biological balance, capable of reshaping how we approach chronic inflammation, infection, and even tissue repair.
The NO–cGMP Axis: A Molecular Bridge Between Circulation and Immunity
At the center of PDE-5 inhibitor pharmacology lies a deceptively simple biochemical cascade. Nitric oxide, synthesized by endothelial and immune cells, diffuses into adjacent tissues and activates soluble guanylate cyclase. This leads to the production of cGMP, a second messenger responsible for smooth muscle relaxation, vasodilation, and intracellular signaling.
Phosphodiesterase-5 serves as the regulatory counterweight. By degrading cGMP, it limits the duration and intensity of nitric oxide signaling. In physiological terms, PDE-5 ensures that vasodilation is controlled and reversible. In pathological states, however, excessive PDE-5 activity may contribute to impaired perfusion and dysfunctional signaling.
Sildenafil and other PDE-5 inhibitors interrupt this regulatory process. By blocking cGMP degradation, they amplify nitric oxide signaling, prolonging vasodilation and enhancing microcirculatory flow. Yet this is only part of the story. cGMP is not merely a vascular mediator—it is also a signaling molecule within immune cells, influencing their activation, differentiation, and cytokine production.
Thus, the NO–cGMP pathway represents a functional bridge between vascular and immune systems. PDE-5 inhibitors do not simply improve blood flow; they modify the biochemical environment in which immune responses are generated and sustained.
Sildenafil and Immune Regulation: Recalibrating the Inflammatory Response
The immune system thrives on balance. Too little activity invites infection; too much leads to tissue damage. Traditional pharmacology often addresses this problem with blunt instruments—either stimulating or suppressing immune function. Sildenafil, intriguingly, appears to operate with greater subtlety.
Experimental findings suggest that PDE-5 inhibition can modulate immune responses by altering intracellular signaling pathways. One key mechanism involves the reduction of myeloid-derived suppressor cells (MDSCs), which are known to inhibit T-cell activity during chronic infections and cancer. By decreasing MDSC-mediated suppression, sildenafil may restore effective immune surveillance.
At the same time, PDE-5 inhibitors can influence cytokine profiles, shifting the balance away from excessive pro-inflammatory signaling. This does not equate to immunosuppression; rather, it reflects a recalibration of immune activity toward a more controlled and effective response.
There is a certain elegance in this mechanism. Instead of forcing the immune system into submission, sildenafil appears to remove inhibitory constraints, allowing physiological processes to function as intended. One might say it behaves less like a hammer and more like a skilled conductor—quietly ensuring that each component plays in harmony.
Vascular Support as an Immunological Strategy
It is tempting to view immunity as a purely cellular phenomenon, but such a perspective overlooks a critical factor: the environment in which immune cells operate. Adequate blood flow is essential for delivering oxygen, nutrients, and immune mediators to tissues. When perfusion is compromised, immune function inevitably suffers.
PDE-5 inhibitors enhance microvascular circulation, improving tissue oxygenation and facilitating the movement of immune cells. This is particularly relevant in conditions characterized by chronic inflammation or infection, where vascular dysfunction often coexists with immune dysregulation.
Improved perfusion also enhances the delivery of pharmacological agents. Antibiotics, for example, rely on adequate blood flow to reach infected tissues. By increasing vascular permeability and flow, sildenafil may indirectly improve the efficacy of antimicrobial therapy.
This dual role—supporting both circulation and immune activity—positions PDE-5 inhibitors as uniquely versatile therapeutic agents. They do not target a single pathway; they optimize the conditions under which multiple physiological systems operate.
Clinical Translation: From Bench to Bedside
The transition from experimental findings to clinical application is rarely straightforward. However, PDE-5 inhibitors offer a distinct advantage: they are already widely used and well-characterized. This provides a strong foundation for exploring new therapeutic indications.
In infectious diseases, sildenafil has been investigated as an adjunct therapy aimed at enhancing immune response and tissue perfusion. Early studies suggest that such an approach may improve outcomes in conditions where chronic inflammation and impaired circulation play a role.
In oncology, PDE-5 inhibitors are being explored for their ability to modulate the tumor microenvironment. By reducing immunosuppressive cell populations and improving vascular function, these agents may enhance the effectiveness of existing treatments, including immunotherapy.
Potential clinical applications include:
- Chronic infections with immune suppression
- Inflammatory diseases involving vascular dysfunction
- Cancer, through modulation of immune evasion mechanisms
- Tissue repair and regeneration
While these applications remain under investigation, the underlying rationale is compelling. Sildenafil is not being repurposed at random; it is being applied in contexts where its fundamental mechanism—enhancing NO–cGMP signaling—addresses key pathophysiological processes.
Limitations and Clinical Realities
Despite the enthusiasm surrounding PDE-5 inhibitors, it is important to maintain a balanced perspective. These agents are not universal solutions, and their effects depend on the underlying physiological context. In patients with intact nitric oxide signaling, the benefits may be modest. In those with significant dysfunction, the impact may be more pronounced.
Adverse effects, though generally mild, must also be considered. Headache, flushing, and hypotension are common, reflecting the systemic vasodilatory action of these drugs. More importantly, contraindications—particularly the use of nitrates—must be strictly observed.
From an immunological standpoint, much remains to be clarified. The immune system is a complex and dynamic network, and interventions that modulate its activity must be approached with caution. Large-scale clinical trials are needed to establish efficacy, optimal dosing, and long-term safety in non-traditional indications.
Nevertheless, the existing safety profile of sildenafil provides a significant advantage. Unlike experimental compounds, it has already undergone extensive clinical evaluation, reducing the barriers to translational research.
Future Perspectives: A New Role for an Old Drug
The expanding understanding of PDE-5 inhibitors reflects a broader shift in medicine toward integrative approaches. Diseases are no longer viewed as isolated malfunctions but as disruptions of interconnected systems. Effective therapies must therefore address multiple dimensions simultaneously.
Sildenafil exemplifies this paradigm. By influencing vascular tone, immune signaling, and tissue environment, it operates at the intersection of several physiological systems. This makes it a valuable tool not only for treating specific conditions but also for restoring systemic balance.
Future research will likely explore combination therapies that leverage these properties. PDE-5 inhibitors may be paired with immunomodulators, antimicrobial agents, or regenerative therapies to achieve synergistic effects. Such strategies represent the next frontier of personalized medicine.
If this trajectory continues, sildenafil may eventually be recognized not merely as a treatment for erectile dysfunction, but as a multifunctional pharmacological platform—a drug that quietly redefined its own identity.
FAQ
1. What are PDE-5 inhibitors and how do they work?
They are drugs that block the enzyme phosphodiesterase-5, preventing the breakdown of cGMP. This enhances nitric oxide signaling, leading to improved blood flow and modulation of cellular processes.
2. How does sildenafil affect the immune system?
Sildenafil influences immune responses by increasing cGMP levels in immune cells, reducing suppressive cell activity and helping restore balanced inflammation.
3. Can PDE-5 inhibitors be used outside erectile dysfunction?
Yes. They are already used in pulmonary hypertension and are being studied for roles in infection, inflammation, and cancer due to their vascular and immunomodulatory effects.
4. Is sildenafil safe for long-term use?
Generally yes, when prescribed appropriately. However, it must not be combined with nitrates and should be used under medical supervision.